Polar flattening in JCTC
published 2015-08-10
Isolated atoms are spheres. One can view them as soft balls which attract each other on long distances and repel each other on short distance. In molecules, however, the shape of atoms is distorted. In fact, it is somewhat difficult to distinguish particular atoms in molecules. The most visible distortion is a flattening of the sphere in the elongation of the covalent bond.
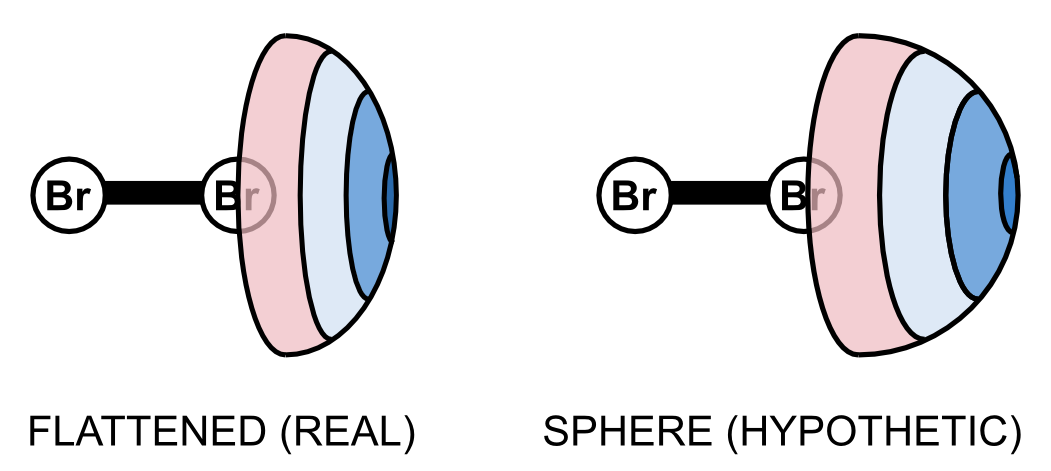
For a long time, I've been interested in halogen bonding - a kind of noncovalent interaction between halogens and electron-rich moieties. In molecules, halogen atoms are also flattened and it is intriguing to know the significance of such distortion. We investigated the geometry of the flattening as well as its consequences on the energetics of halogen-bonded complexes. The calculations were done by Robo Sedlák. The results show that the magnitude of the flattening depends on the electron density surface and that the stabilization energy is affected by up to 15%. In other words, if the halogens in molecules were spherical, the strength of their halogen bonds would be lower by up to 15%. Recently, our conclusions were published in the Journal of Chemical Theory and Computation.
